
The relative amount of sugar in a beverage determines its sweetness (see Figure 3.14). The relative amount of the active ingredient in a medicine determines its effectiveness in achieving the desired pharmacological effect. The relative amounts of iron, carbon, nickel, and other elements in steel (a mixture known as an “alloy”) determine its physical strength and resistance to corrosion. The relative amount of oxygen in a planet’s atmosphere determines its ability to sustain aerobic life. Similar to a pure substance, the relative composition of a mixture plays an important role in determining its properties. However, mixtures-samples of matter containing two or more substances physically combined-are more commonly encountered in nature than are pure substances. Preceding sections of this chapter focused on the composition of substances: samples of matter that contain only one type of element or compound. Perform dilution calculations using the dilution equation.Calculate solution concentrations using molarity.Describe the fundamental properties of solutions. 
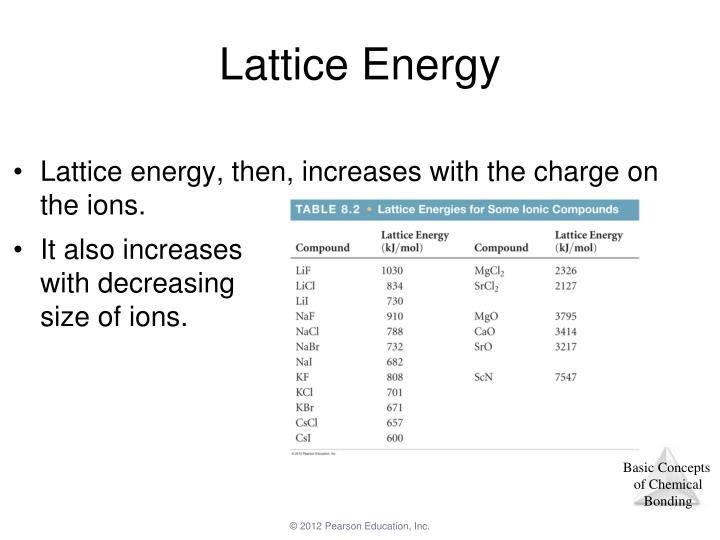
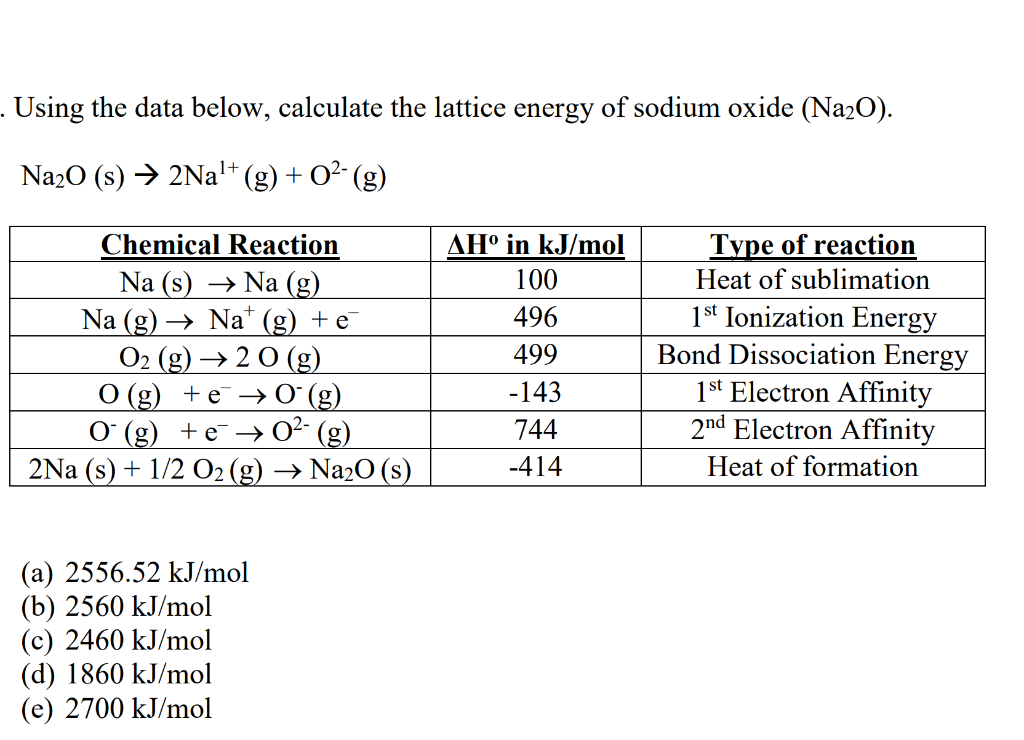
The calculation is regarding the lattice energies and values which when acquired through this method are called the theoretical values.By the end of this section, you will be able to: This method focuses on how much energy would be released to dissociate the atom from solid to gaseous. Lattice enthalpies acquired through this procedure are called the experimental values. In the Born Haber Cycle method which is also known as the Hess Law Cycle the changes in the enthalpy can be measured. The first method is known as the Hess Law cycle or Born Haber cycle and the second method is called physics style. The lattice enthalpy could be calculated in two ways. When the energy is required to form lattice from gaseous ions it is known as lattice formation enthalpy which is always negative.įor example, The lattice formation enthalpy of NaCl is -787 KJ mol -1 When energy is required to separate 1 mole of a solid crystal into gaseous ions known as lattice dissociation enthalpies which is always positiveįor example, the lattice dissociation enthalpies for Nacl is +787 KJ mol -1 Lattice enthalpy can be described in two ways, formation of solid compounds from gaseous ions and separating solid into gaseous ions. Is Lattice Enthalpy Negative or Positive? Size of the Atom:Ī small atom has a small interatomic distance between them and they have a stronger binding force between them which requires a higher lattice enthalpy.įor example: the ions present in magnesium ions are small which means the bond between them is strong and thus requires higher lattice enthalpy The force of lattice crystal is directly proportional to the magnitude of charge which means the higher the magnitude of charge higher the would-be force and vice versa.įor example, potassium chloride and calcium have the same amount of lattice crystal but calcium has greater force because calcium ions have +2 charges whereas sodium chloride has +1 and as we know the greater the force this calcium has more electrostatic force than sodium chloride. There are two factors that affects the lattice enthalpy are charge of ion and size of the atom which are explained below: Charge of Ion:įorce is present in ions of lattice crystals, due which ions are attracted to each other. The addition of enthalpy to an electron can be explained as the amount of energy exerted when an atom gains an electron, hence greater the electron affinity, the larger will be the lattice energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
